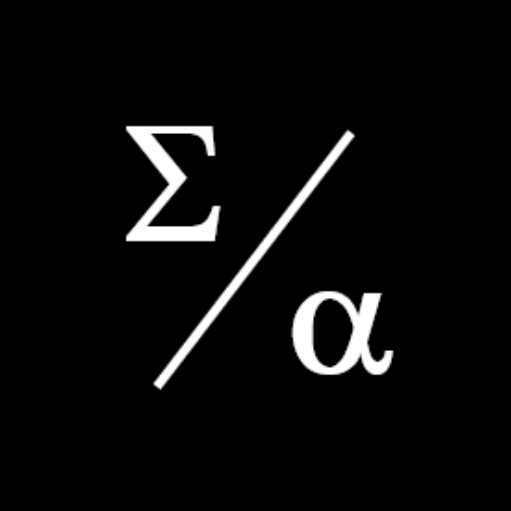Many bioentrepreneurs incorrectly estimate the value of their technology by failing to account adequately for the cost, risk, and time inherent in product development.
Venture capitalists are often wary of investing in biotechnology because bioentrepreneurs seldom provide realistic estimates of the value of their technologies.
To evaluate a new biotechnology, an entrepreneur must account for the future revenue from the final product, the cost and time needed to get the product to market, and the various risks faced along the way.
Entrepreneurs can approach the venture community with a more rational basis for investment by expressing biotechnology in terms of risk-adjusted net present value (rNPV; see “Glossary”), as discussed here. Investments, milestone payments, clinical trial costs, and royalties on sales can then be compared directly using the common currency of rNPV.
A researcher has made a scientific breakthrough that could be worth millions of dollars. To attract the investment needed to commercialize biotechnology, the researcher must now convince venture capitalists and pharmaceutical companies of its potential. However, investors want to know what biotechnology is worth today and will require evidence to substantiate this estimate.
The numbers game
Unfortunately, estimates of the value of a biotechnology are all too often clearly unrealistic. “Valuations” are typically made in the following (unrealistic) manner: “The market for our product is $2 billion per year, so if we capture only 10% of that market for 10 years, then the company is worth $2 billion today, less development costs.” Perhaps as a result, the venture capital community often judges a company on the basis of its management’s expertise rather than the underlying asset of real value—the biotechnology.
How, then, can we put a price tag on biotechnology? The best solution is to evaluate biotechnology by estimating the rNPV. Using rNPV, researchers and potential investors can price the biotechnologies that they are considering selling, investing in, or acquiring. However, it should be noted that the management, science, and intellectual property surrounding a biotechnology must all be of the highest quality to interest the venture community; if any of these are seriously lacking, the biotechnology is effectively worthless.
Start at the end
The first place to start when valuing biotechnology is at the end—the projected revenue stream. The end product for most biotechnologies is a medicine, and the payoff is frequently the royalty due the biotechnology company paid from the estimated annual revenue of the product sold by a manufacturing and marketing partner (or sales of the product, if the company retains all rights). In general, annual revenues of a product are estimated using the current sales of drugs used to treat similar indications. As discussed previously1, the take-home percentage (typically divided between milestone payments and royalties on gross sales) due pre-market biotechnology developers is about 40% of gross product revenue (see “Parameters for biotechnology”).
To illustrate the rNPV method, we have created a hypothetical scenario: A company has developed Acmed, a potential treatment for asthma. The preclinical science and intellectual property are sound, and Acmed has passed initial testing in animals and is now ready to enter phase 1 trials. The company is seeking venture funding and partnering opportunities with multinational pharmaceutical companies, so what should they charge for Acmed today?
The annual market for asthma treatments is around $5.8 billion. To estimate Acmed’s market share, the product is compared with other asthma medications on the market. Competition within the asthma market is intense, and the anticipated market share for Acmed may be just 5%—a “moderate to small” share on the spectrum of market shares currently captured by pharmaceutical companies. The annual gross return of Acmed will therefore be about $290 million. Of this sum, 60% is reserved for the eventual marketing and manufacturing partner, and 5% is reserved as a royalty for the university that invented Acmed. This leaves 35%, or an annual return of about $100 million, as the royalty due the biotechnology company that develops Acmed through pre-market research and development stages.
Consultation with a patent attorney suggests that Acmed will be defended from competition for the next 18 years. The payoff for Acmed is, therefore, $100 million a year for 18 years minus the years that it takes to get the product to market. It should take eight years to carry out clinical trials and have the drug approved by the US Food and Drug Administration (FDA), and so Acmed’s potential payoff for the biotechnology company is $1 billion (see “Acmed Payoff”).
Although we have identified the theoretical payoff, the true value of Acmed is far less. Several factors consume the present value of the biotechnology in nibbles, bites, and chomps. Indeed, these factors can eat up the entire value of the biotechnology—leaving nothing for the biotechnology company or its investors. These three factors are the cost, risk, and time associated with drug development.
Factor 1—Cost
The cost of drug development can be estimated using industry standards2, 3, and any deviations from these standards must be justified. Acmed’s development incurs the costs associated with additional animal studies, clinical trials, and filings to the FDA. By comparing with clinical data from currently marketed asthma drugs, it is possible to estimate how many subjects will need to be enrolled in clinical trials. Clinical trials involving asthma inhalants such as Acmed are data-intensive because multiple tests are performed over a relatively extended time period, and the trials will be conducted in the United States, so the costs for each subject will be at the top end of the range.
Overhead costs vary considerably between companies, and the value of the technology will vary in parallel. The same situation arises in other walks of life: For example, if you can repair your own house, total repair costs are lower, and the house is effectively worth more to you than it would have been to an unskilled owner. However, in this example we have left out the “overheads” and estimate Acmed’s intrinsic value. The total cost of developing Acmed is $23 million (see “Acmed costs”).
Factor 2—Risk
It would be grossly inappropriate simply to subtract the costs from the payoff to estimate Acmed’s intrinsic value. Such a calculation would imply that each clinical trial was a guaranteed success. Instead, clinical drug development should be regarded as a series of high-risk wagers where success in the first wager (e.g., a phase 1 trial) allows a company to make additional wagers (e.g., phase 2 and 3 trials) before reaching the ultimate payoff (e.g., a marketed drug). A company may never see the payoff, but then the company may not have to pay for a phase 3 trial. Each wager is associated with an ante (the stake or sum wagered), such as the cost of each clinical trial. The key to determining the value of the wager series is to risk-adjust both the payoff and the ante (see “Risk adjustment”).
Acmed appears to be a typical pharmaceutical and is estimated to be associated with normal development risks. Each of Acmed’s costs are risk-adjusted by the risk inherent to each stage (see “Risk-adjusted Acmed costs”). These risk-adjusted costs are then subtracted from the risk-adjusted payoff. Acmed’s risk-adjusted costs are $8.9 million. Acmed’s risk-adjusted payoff is $200 million, and so if all sales and pre-market stages were completed instantaneously, the resultant risk-adjusted value of Acmed would be about $191 million.
Factor 3—Time
A company would rather have a dollar today than a dollar tomorrow because today’s dollar can be invested and earn a return, increasing its worth tomorrow. By the same argument, a dollar received tomorrow is worth less than a dollar received today. The net present value (NPV; see “Glossary”)—a standard finance equation—is what tomorrow’s cash flow would be worth today.
The amount that future money loses in value each year is termed the “discount rate”. Discount rates normally include many factors including risk. However, in the Acmed example, the discount rate is independent of R&D risk. We assume here that the discount rate is equivalent to the 20% internal rate of return generally expected by the primary sources of capital available to biotechnology companies—venture capitalists and large pharmaceutical companies1. Research and development (R&D) risk is accounted separately by development stage.
The effect of discounting can be dramatic. For example, if clinical trials began today, Acmed would not begin earning revenue for another nine years. Furthermore, the $1 billion in total revenue generated is spread out over 10 years (Acmed’s has only 18 years of blocking patent life remaining). Assuming a 20% discount rate, the NPV of Acmed’s payoff cash flow is only $117 million total (calculation not shown), and this is before any adjustment has been made for development risks. Because the payoff will not come for some time, the NPV of the money is much lower than one might have expected. Clearly, time is a significant factor when valuing biotechnology, especially when the brunt of clinical trial costs comes before revenue is generated. On the upside, the most expensive clinical trials take place later in development and so have significantly discounted NPV. In the case of Acmed, discounting reduces the pre-revenue costs of Acmed from $23 million to a present value of $12.6 million (calculation not shown).
rNPV
To calculate the true present value of biotechnologies, revenue, cost, risk, and time must be combined into a single calculation of rNPV. In the rNPV equation, Equation (2), the present value of each risk-adjusted cost is subtracted from the present value of the risk-adjusted payoff to arrive at the rNPV of the biotechnology.
By adding together all of Acmed’s costs and risks and then discounting for time, the true rNPV is finally revealed. Today, Acmed is worth about $18 million (see “Acmed’s rNPV“).
Investment
Estimates of rNPV can be useful in deal-making scenarios: For example, if a company wants to raise money from investors, how much of its equity is it fair to give away in return? If a pharmaceutical company wants to pay milestones and a royalty on sales, what should this royalty be? Both investments and milestone payments can be calculated simply by reducing each to the common currency of the rNPV.
For example, a venture capital company is willing to invest $9 million in Acmed. Today’s $9 million investment has an rNPV of $9 million, which is added to Acmed’s rNPV ($18 million) to yield a new rNPV of $27 million. The venture capital contribution represents a third of the assets of the now-capitalized project, so a fair value for the venture capital investment would be about 33% of Acmed. (Although we will not develop this method here, the equity must be increased to account for company overheads and anticipated equity dilutions.)
In a second scenario, a pharmaceutical company is willing to in-license Acmed for milestone payments of $5 million today, $10 million on entering phase 2, $15 million on entering phase 3, and a royalty on gross sales. Also, the pharmaceutical company will split Acmed’s remaining development costs. What would be a fair royalty?
By calculating the rNPV of each milestone and the clinical trial costs borne by the pharmaceutical company, the pharmaceutical company has made an investment with an rNPV of $15.9 million. In return, it would be fair to give the pharmaceutical company 68% of the $23.4 million rNPV of Acmed’s payoff. Acmed’s developers would retain 32% of the 35% R&D royalty on Acmed’s gross revenue—about an 11% royalty.
Selling price versus fair value
Using the rNPV, the inventor and investor can arrive at a realistic value of a biotechnology (see Fig. 1). By adopting an auditable valuation approach, biotechnology companies may be able to seek debt financing even at early R&D stages. However, as Steven Burrill, chief executive officer of Burrill & Company (San Francisco, CA) cautions:”Notwithstanding all the fancy math, the real way these tech companies are valued is based on comparables … the real value is determined on an arm’s-length negotiation.” Even so, knowing the underlying value of a biotechnology can be critical for getting the best deal from either side of the negotiation table. The same applies when buying or selling a house: You get the best deal when you know the house’s value based on an accurate appraisal. Likewise, you can set an advantageous price by knowing the fair value of the biotechnologies—the rNPV.
Figure 1: The value of biotechnology.
Simplistic cash flows (in red), which include revenue and costs, present unrealistically high valuations for biotechnologies. A better representation is the net present value (NPV; in green), which discounts the revenue cash flow over time, but even the NPV overestimates the value of biotechnologies during all R&D stages. Risk is mitigated as biotechnologies progress through development. When this increasingly mitigated risk is taken into account, the risk-adjusted cash flow can be discounted to arrive at the risk-adjusted NPV (rNPV; in blue). The rNPV is an estimate of the fair price of a biotechnology. Note that rNPV coincides with NPV only once risk is mitigated.
© Amy Center
Note: A Microsoft Excel spreadsheet for calculating the rNPV is available as supplementary information.
The spreadsheet version accounts costs by calculating the risk-added costs rather than risk-adjusted costs. Risk-added costs are Ci/Ri; R0 is multiplied later to arrive at the risk-adjusted costs. This rearrangement of the equation yields the same rNPV.
Acknowledgements
We thank D. Constable of Hollister–Stier Laboratories (Spokane, WA), S. Litwin of the Fox Chase Cancer Center (Philadelphia, PA), M. Sanders of ProPharma Partners (Hayward, CA), S. Burrill of Burrill & Company (San Francisco, CA), and S. Trimbath and P. Wong of the Milken Institute (Santa Monica, CA), whose input was invaluable; we also thank J. Wadsack of the New Jersey Virtual Campus (Chatham, NJ) and J. Johnson (Moscow, ID), without whose support this publication would not have been possible.
Jeffrey J. Stewart1, Peter N. Allison1 & Ronald S. Johnson1
Jeffrey J. Stewart (e-mail: jjs@alumni.princeton.edu), Peter N. Allison, and Ronald S. Johnson are with the life sciences investment banking firm, BioGenetic Ventures (Bellevue, WA).
References
- Moscho, A. et al. Deals that make sense. Nat. Biotechnol. 18, 719–722 (2000). | Article | PubMed | ISI | ChemPort |
- US Congress, Office of Technology Assessment. Pharmaceutical R&D: costs, risks and rewards, OTA-H-522. (US Government Printing Office, Washington, DC; February 1993).
- PhRMA. The pharmaceutical industry profile 2000. Pharmaceutical Research and Manufacturers of America. http://www.phrma.org/publications/publications/profile00/
- US Food and Drug Information Office of Planning. FY 2000 Performance Report to Congress for the Prescription Drug User Fee Act of 1992 as reauthorized and amended by the Food and Drug Administration Modernization Act of 1997 (2001). http://www.fda.gov/ope/pdufa/report2000/default.htm
Source
http://www.nature.com/bioent/2003/030101/full/nbt0901-813.html